Guideline for Use of Octreotide in Fistula Management
exp date isn't null, but text field is
Objectives
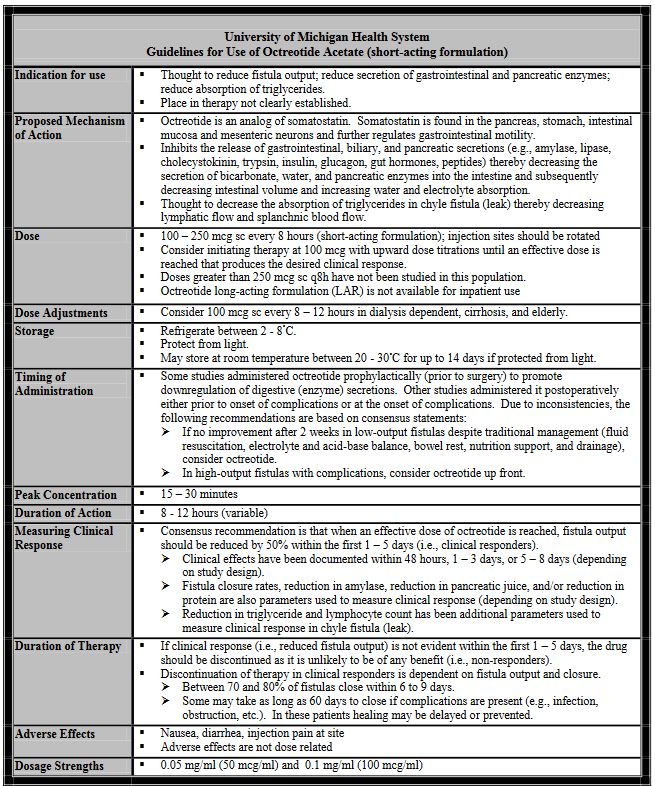
To outline criteria for use of octreotide acetate (short-acting formulation) to reduce fistula output and secretion of gastrointestinal and pancreatic enzymes and potentially accelerate fistula closure.
Guideline
Octreotide short-acting formulation may be initiated by a health care professional for fistula management according to the criteria defined in this guideline. Octreotide long-acting formulation (LAR) is not available for inpatient use. Prescribers will be required to document that octreotide use is for a high-output fistula. Fistula output must be assessed within the first 1-5 days of therapy. If a 50% decrease in fistula output is not seen octreotide therapy should be discontinued.
Background:
A fistula is an abnormal contact between two epithelialized surfaces. These surfaces can include the skin, a part of the gastrointestinal tract, or another organ system (e.g., biliary, respiratory, or genitourinary). For example, an enterocutaneous fistula (ECF) involves an abnormal communication between the bowel and skin. Fistulas can range from being undetected and causing few symptoms to being life-threatening. Most develop as a complication of surgery or trauma (e.g., anastomotic leakage, pancreatic fistulas). For example, pancreaticoduodenectomy surgery (Whipple’s procedure) alters the anatomical organization of the pancreas and intestines, potentially creating an abnormal contact between (leakage of) pancreatic secretions and gastrointestinal enzymes.
A chyle fistula (leak) may develop as a result of lymphatic injury from trauma or surgery in the chest, abdomen, or neck (thoracic duct).Other fistulas may begin as an inflammation that moves into the muscle tissue and develops into an abscess. The abscess could develop into a fistula, which degrades the muscle until an opening in the skin is created. The severity of a fistula(s) correlates with the volume and enzymatic composition of its output, which is further dependent on its anatomical location.
In a healthy patient with an intact functioning intestine, 8-10 L of fluid flows through the jejunum daily. This is comprised of oral intake, 1.5 L from the salivary and gastric glands, 1 L from the pancreas, 1 L from the bile, 1.8 L from the small intestine, and 200 ml from the duodenal Brunner’s gland. Only 100 –200 ml of this total volume is excreted in the stool while 98% is reabsorbed proximal to the ileocecal valve.
In addition, enzymes, nutrients, and electrolytes accompany fluid excretion and reabsorption. Enzymes can include gut hormones (e.g., gastric acid secretion, histamine, somatostatin, CCK), pancreatic enzymes (e.g., amylase, lipase), and salivary amylase and gastric pepsin. Leakage of enzymes can have a corrosive effect and digest surrounding tissue(s), thereby contributing to fistula formation or exacerbation.
In general, all fistulas cause loss of fluid, electrolytes, nutrients, and enzymes. Therefore, progression of a fistula(s) or inadequate management can lead to fluid and electrolyte losses (i.e., dehydration); nutrient losses (i.e., malnutrition); enzyme losses causing inflammation and excoriation of previously normal skin (i.e., delay in wound closure); changes in intestinal motility and secretion (depending on anatomical location); abscess formation; sepsis.
Although studies have correlated high-output fistulas with an increase in morbidity and mortality, it is not a clear prognostic indicator of fistula closure. The literature uses the following parameters to define output:
- High pancreatic fistula output> 200 ml/24 hours
- High intestinal fistula output> 500 ml/24 hours
- Low pancreatic fistula output< 200 ml/24 hours
- Low intestinal fistula output< 500 ml/24 hours
The goal of fistula management is to create an environment that promotes fistula closure but also optimizes the patient’s overall health status.
Fistula management is guided by volume of output and anatomical location. Fistula output is measured and managed via the placement of surgical drains (e.g., JP drains).
Best practice (i.e., traditional management) includes adequate fluid resuscitation, electrolyte replacement, bowel rest in conjunction with nutritional support, and fistula drainage.
Nutritional support and bowel rest help to reduce the stimulation and secretion of enzymes in the discharge, which improves time to wound healing and helps reduce output. Ultimately, fistulas either close spontaneously or require surgical closure.
Pharmacological therapy has been explored to reduce output and secretion of enzymes and/or to modify the composition of gastrointestinal and pancreatic secretions. However, validation on use of pharmacological agents is lacking. Potential strategies include the following and consideration of use is dependent on anatomical location:
- Anti-secretory agents/reduction of gastric acid (proton pump inhibitors, histamine blockers, octreotide)
- Anti-motility agents (loperamide, imodium, codeine)
- Pancreatic enzymes (pancrelipase)
- Bile salt loss agents (cholestyramine)
Rationale for octreotide acetate in the management of fistula(s):
The efficacy of octreotide in prevention and treatment of gastrointestinal, pancreatic, and chyle fistulas remains unclear (i.e., to reduce fistula output and/or secretion of gastrointestinal and pancreatic enzymes).
Although studies have shown it to reduce output in established fistulas, differences in study designs (e.g., primary endpoints, dosing, timing of administration, treatment versus prophylaxis) make it difficult to draw any firm conclusions regarding its place in therapy. Furthermore, there is insufficient evidence to show that octreotide reduces fistula closure rates, time to closure, or overall complications.
Consensus recommendations state that octreotide is a relatively safe drug that maybe beneficial if the prescriber believes that a reduction in output may facilitate overall management. It is important to note that only the short acting formulation of the drug has been utilized in clinical trials; there are no data assessing the efficacy of the long-acting (LAR) formulation.